
Our Femtech Horizon Maps highlight technologies across women's health, showing where innovation is emerging and where gaps remain. By visualizing the full continuum of care for specific verticals, these maps help founders, clinicians, and investors identify opportunities, address unmet needs, and build more integrated solutions.
This Breast Health map examines innovation across the care journey, from early awareness through screening, intervention, and long-term survivorship.
When we talk about 'breast health' innovation, what we are largely talking about is breast cancer.
The overwhelming majority of technologies in this landscape are built around cancer detection, diagnosis, treatment, and survivorship. Far fewer solutions address non-cancer breast conditions such as benign breast disease, chronic breast pain, fibrocystic changes, cosmetic or reconstructive decision support, or broader breast wellness. The category is labeled 'breast health,' but structurally it functions as a breast cancer innovation ecosystem.
Within that ecosystem, breast cancer screening is one of the most established programs in modern medicine. Mammography is standard of care and nearly universally available at health systems across the US, yet only around 50–60% of eligible women are current with screening, depending on the study and population examined. And the experience for patients post diagnosis also remains fragmented and frustrating.
Women move from screening to follow-up imaging, to biopsy, to surgery, to oncology, to radiation, to reconstruction, and into survivorship. Each step is often managed in a separate system with limited data continuity. Risk assessment is frequently episodic rather than longitudinal. Survivorship support varies dramatically by geography and institution.
Innovation is accelerating across breast health, but it is uneven. Some segments are highly saturated, while others remain structurally underbuilt. This map visualizes the imbalance.
Innovation spans the full continuum of care. However, activity concentrates heavily in screening and diagnostic technologies, while survivorship and longitudinal monitoring receive comparatively less attention.
For this map, we examined four phases of clinical need:
Each phase reveals a distinct pattern of innovation and a distinct set of gaps.
Awareness in breast health increasingly blends education with risk understanding and screening navigation.
Within this phase, innovation primarily draws from:
Risk Prediction & Preventive Stratification Platforms, which provide structured questionnaires and risk scoring tools that prompt earlier screening conversations.
Workflow & Operational Infrastructure, when used to guide referral pathways or standardize intake and screening recommendations.
Some tools in this category look less like general education and more like structured decision support. For example, interactive risk assessment experiences can help move people from awareness into appropriate screening conversations.
The opportunity at this stage is continuity. Awareness tools can start the momentum, but accessible screening has remained a point of friction — particularly for women in areas with limited imaging infrastructure, and for those in primary care roles that leave little time for their own preventive healthcare. Fortunately, innovation in self-screening and diagnostic kits may help reduce that friction.
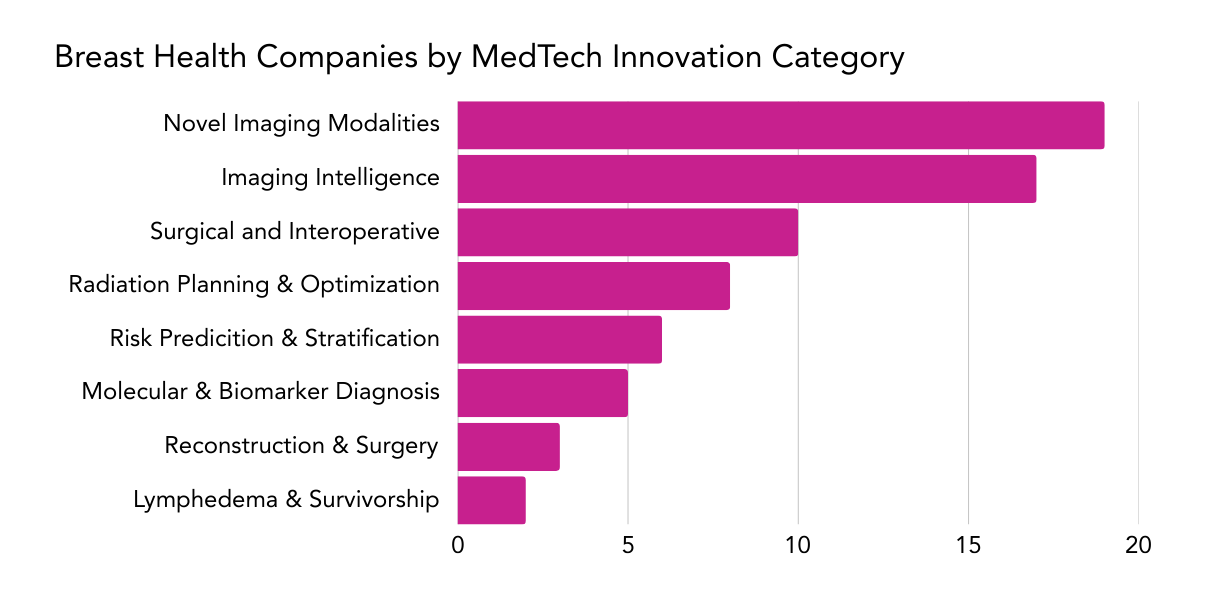
Screening and diagnosis represent the most active area of innovation in breast health.
This phase is shaped by four primary innovation categories:
Imaging Intelligence (AI layered on existing modalities), which enhances mammography, tomosynthesis, MRI, and ultrasound through detection algorithms, density automation, and triage systems.
Novel Imaging Modalities, including microwave, optical, spectral, and other hardware-based approaches that attempt to improve performance in dense tissue or reduce radiation exposure.
Risk Prediction & Preventive Stratification Platforms, which integrate imaging features, genetics, and patient history to estimate five-year or lifetime risk and guide screening intervals.
Molecular & Biomarker Diagnostics, which move beyond image interpretation and attempt to detect or monitor cancer through biological signals.
Workflow & Operational Infrastructure also plays a supporting role in this phase by improving reporting, quality assurance, and system integration inside radiology environments.
Overall, screening innovation is robust, but it is heavily weighted toward optimizing existing imaging pathways.
Innovation in treatment focuses primarily on procedural precision and delivery optimization.
This phase is driven by:
Unlike screening, where software overlays dominate, treatment innovation centers more on hardware systems, procedural tools, and intraoperative guidance.
The trend here is toward precision and functional restoration. In reconstruction specifically, the most interesting signals are technologies that shift recovery and control back to the patient, and research that treats sensation as a first-class outcome rather than an afterthought.
Maintenance and survivorship represent the smallest but arguably most underbuilt phase of the continuum.
Innovation here primarily reflects:
Lymphedema Monitoring & Survivorship Tools, which address post-treatment complications and long-term symptom tracking.
Workflow & Operational Infrastructure, when used to coordinate follow-up, monitor outcomes, or structure survivorship programs.
Some companies address post-treatment complications through device-based monitoring — for example, Cacto Health offers remote monitoring specifically for lymphedema.
While several broader oncology platforms offer survivorship features that can be applied to breast cancer patients, very few platforms are purpose-built for breast cancer survivorship. There is a meaningful distinction between platforms designed for broad oncology monitoring that can be extended into breast programs, and platforms that build breast-specific workflows, symptom sets, and longitudinal risk frameworks from the ground up. The latter category remains largely vacant.
Examples of broader oncology survivorship platforms that include breast cancer use cases:
These platforms represent meaningful progress in structured survivorship care. However, they are not breast-specific, and the breast cancer survivorship population — which numbers in the millions in the US alone — currently lacks dedicated remote patient monitoring (RPM) or remote therapeutic monitoring (RTM) infrastructure built around its specific clinical needs. RPM covers physiologic data collection; RTM covers therapeutic adherence and symptom response. Neither is currently well-represented in breast-specific survivorship platforms.
Given the size of the survivorship population, this represents a structural gap.
Three themes emerge clearly across the breast health landscape:
Screening remains the center of gravity. Innovation is concentrated in imaging intelligence and hardware improvements, reinforcing mammography-based pathways. Breast imaging and related diagnostic technologies also represent one of the most regulatorily mature segments in women's health, accounting for a substantial share of FDA-cleared femtech devices compared to other verticals.
Biology-first approaches are present, but still a thinner layer. A smaller set of companies are exploring biological signals, pathology intelligence, and non-imaging approaches that could expand what early detection looks like.
Survivorship-specific infrastructure is underbuilt. Remote monitoring and longitudinal care models exist, but few are purpose-built for breast cancer populations.
The next phase of breast health innovation will likely depend less on incremental image interpretation improvements and more on connecting risk assessment, imaging, biological signals, treatment data, and long-term monitoring into cohesive systems.
The map makes four opportunity areas especially clear. These examples illustrate where innovation is pushing beyond incremental optimization and toward structural change.
Most screening innovation still assumes imaging is the starting line. A smaller set of approaches attempt to detect cancer by reading biological signals directly.
Detection is only the first chapter. Monitoring for molecular residual disease and early relapse is emerging as a critical layer.
Survivorship is where patients spend years, yet longitudinal infrastructure built specifically for breast cancer remains thin. See the Maintenance & Support section above for a full breakdown of current platforms and the gap that remains.
Risk is often treated as a one-time intake step. A newer class of tools pushes toward lifetime stratification that meaningfully changes screening intensity.
Several structural gaps persist:
Women should not have to navigate a fragmented system at the most vulnerable points in a cancer journey. We hope to see a more unified continuum of care where breast health is treated as a continuous, longitudinal experience, from first screening through years of survivorship, rather than a series of disconnected clinical encounters.
Sources